Kamilah Hauptmann
Shitpost Sommelier
- Joined
- Sep 20, 2018
- Messages
- 15,081
- Location
- Cat Country (Can't Stop Here)

- SL Rez
- 2005
- Joined SLU
- Reluctantly
Post lifted from a lolyer elsewhere:
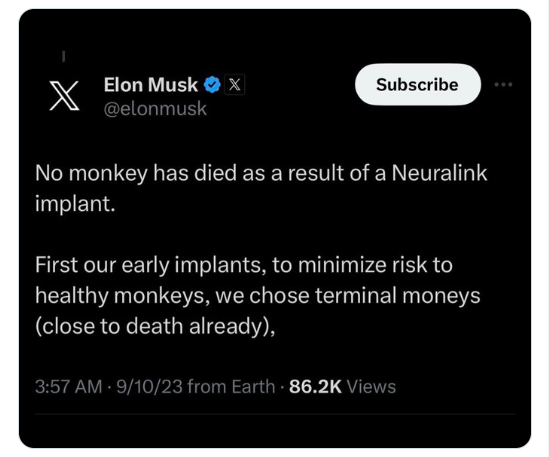
Nah, Elon is lying. There is no regulatory basis for using a decedent model as submission-enabling pre-clinical research - it is essentially basic research that is exceptionally useful in xenotransplantation, where substantial differences in human vs. non-human primate immune response may make a non-human primate model less useful in early discovery. At present, that kind of data cannot be used to enable systematic human clinical investigations. Non-human primate work is still required.
For an investigational device like Neuralink, you need high quality animal model data (most likely non-human primate at some point) to obtain an investigational device exemption and advance to human clinical trials. Using moribund animals is a serious experimental confound. There is no such thing as compassionate use or phase 1b study - where you would study the device in persons with a particular disease state alongside the standard of care - for animals. I would also be very surprised if any IACUC (Institutional Animal Care and Use Committee that has oversight of animal studies) would sign off on such research. Research animals with terminal illnesses are euthanized. Maintaining research animals that are "terminal" is likely unethical.
So, if this were true (it is not), it would likely either (1) tank the utility, such as it was, of the referenced non-human primate pre-clinical work using the "terminal" animals or (2) get the responsible IACUC and investigators inspected and audited by USDA, because it reads like a violation of the Animal Welfare Act.
This is just another thing he made up.